Development in the Concept of Bacterial Polysaccharide Repeating Unit-Based Antibacterial Conjugate Vaccines | ACS Infectious Diseases

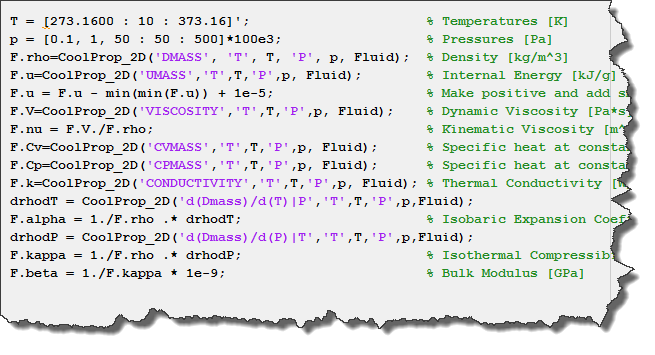
A class diagram of (a) the thermophysicalModels library with critical... | Download Scientific Diagram

Energy Conversion CHE 450/550. Ideal Gas Basics and Heat Capacities - I Ideal gas: – a theoretical gas composed of a set of non-interacting point particles. - ppt download

Cardiac toxicities in multiple myeloma: an updated and a deeper look into the effect of different medications and novel therapies | Blood Cancer Journal

The equation is given below, CP=CV+TV(Beta2/K), links CP and CV with Beta and K. Use this equation to evaluate CP - CV for an ideal gas. CP=CV+T(Lambda P Lambda T)V(Lambda V Lambda

Cp-Cv for real gas, in terms of alpha & beta, change in internal energy with respect to volume - YouTube
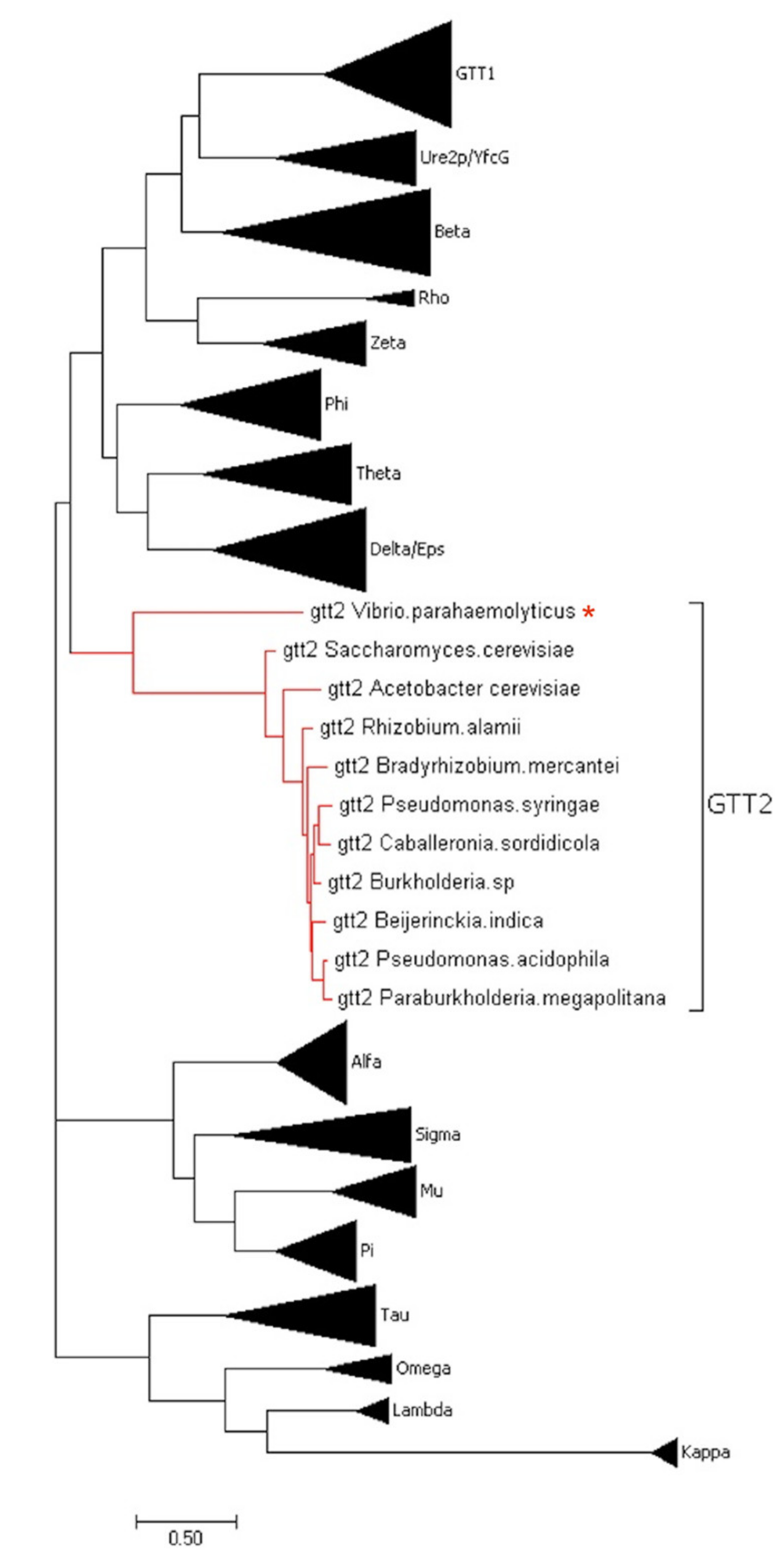
Toxins | Free Full-Text | A Novel Glutathione S-Transferase Gtt2 Class (VpGSTT2) Is Found in the Genome of the AHPND/EMS Vibrio parahaemolyticus Shrimp Pathogen
Ideal and Real Discharge Coefficients – Using Fundamental Equations of State in Mass-Flow Measurements with Sonic Nozzles R. S

If Cp and Cy denote the specific heats of nitrogen per unit mass constant pressure and constant volume respectively, then Cp - Cv = 28R Cp - CV =R/28 Cp - Cy=R/14 •